No products in the basket.
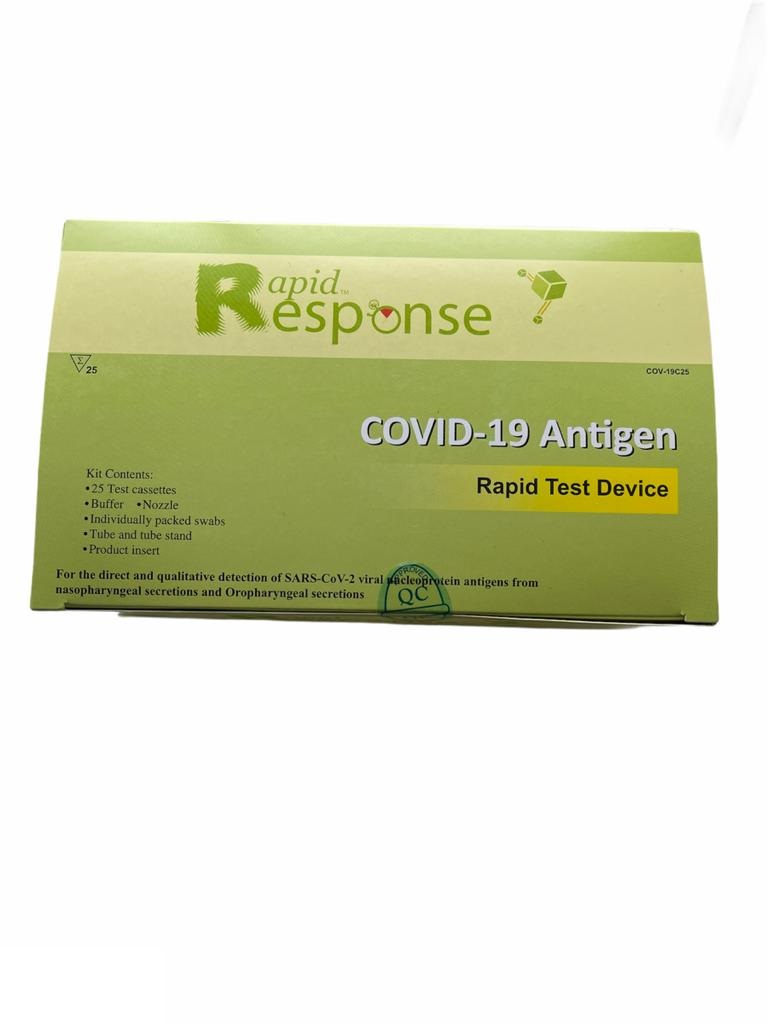
Rapid Response COVID-19 Antigen Test Kits (Pack of 25)
£425.00
Sample Nasal / Nasopharyngeal / Oropharyngeal secretions
Format Cassette
Quantity 25 Tests/Kit
Results in 15 minutes
Storage Condition 2-30°C/36-86°F
Test Principle Immunochromatographic Assay
CONTENTS
Individually packed test devices
Extraction Buffer
Extraction tube
Nozzle with filter
Tube stand
Individually packed swabs
Package Insert