No products in the basket.
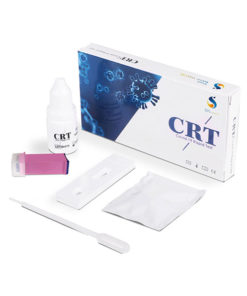
CAT Rapid Lateral Flow Antigen Test Kits (Pack of 10)
£369.99 £289.00
Throat and Nose Swab Test 15 Minute Detection Time
- Accuracy 98.10%
- Sensitivity 97.59%
- Specificity 98.43%
- CE certified
- Sample from the throat and nose
- Fast, accurate results
- Fully sealed for safe waste
- Can be used at home or point of care
- Video guide
SKU: 10TKIT
Categories: ANTIGEN TEST KITS, Coronavirus
Throat and Nose Swab Test. 15 Minute Detection Time
2019-nCoV Rapid Lateral Flow Antigen Test Kit is an immunochromatography based one step in vitro test. It is designed for the rapid qualitative determination of SARS-CoV-2 virus antigen in nasopharyngeal swab specimens. 2019-nCoV Rapid Lateral Flow Antigen Test Kit cannot be used as the basis to diagnose or exclude SARS-CoV-2 infection. This item should be administered by a healthcare professional.
SUMMARY
COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection, asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is J to 14 days, mostly 3 to 7 days. The main symptoms include fever, fatigue, and dry cough. Nasal congestion, runny nose, sore throat, myalgia, and diarrhoea are found in a few cases.
PRINCIPLE
2019-nCoV Rapid Lateral Flow Antigen Test is an immunochromatographic lateral flow device that employs the principle of double antibody sandwich method. Colloidal gold conjugated ant -SARS- CoV-2 antibodies are dry-mobilized on the test device. When the specimen is added, it migrates by capillary diffusion through the strip to re-hydrate the gold conjugate complexes. If present at or above the limit of detection on, SARS-CoV-2 viral antigens will react with the gold conjugate complexes to form particles, which will continue to migrate along the strip until the Test Zone (TJ where they are captured by the immobilized ant -SARS-CoV-2 antibodies to form a visible red line. If there are no SARS-CoV-2 viral antigens in the spec men, no red line will appear in the Test Zone IT). The gold conjugate complexes will continue to m grate alone until being captured by immobilized antibody in the Control Zone (CJ to form a red line, which indicates the valid ty of the test.